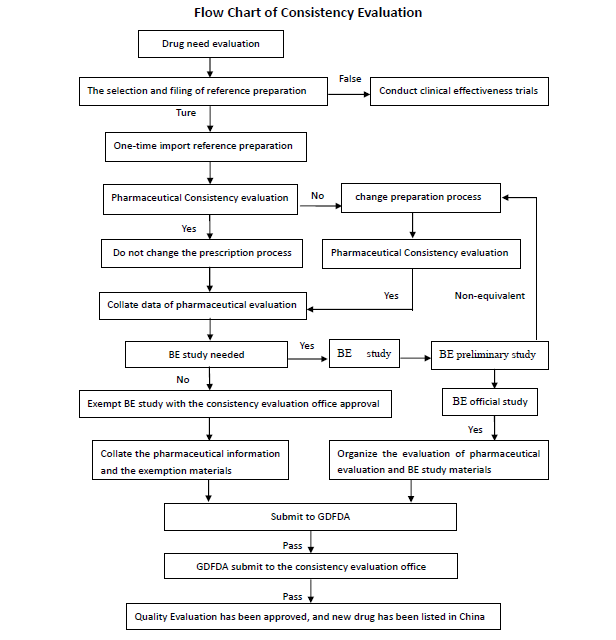
RESEARCH SERVICES:
(1) Research on preparation process or variation.
(2) Preparation process development, Pilot magnification, and process validation.
(3) Research on impurities and original comparison, impurity profiling, etc.
(4) Revelopment and verification for assay and related substance.
(5) Research on dissolution rate and in vitro consistency evaluation.
(6) Write the submission documentation, assist the manufacturer in on-site inspection and registration.
BE STUDY SERVICES:
(1) Provide the design of the BE study program.
(2) Application for ethics report and subject enrollment.
(3) BE preliminary study.
(4) BE study.
(5) Establish bio-sample test method and verification.
(6) Write BE study report.